
About
Reina Mebius is professor in Molecular Cell Biology at the Department of Molecular Cell Biology & Immunology. She has contributed to the field of lymphoid organ development showing that communication between stromal cells and immune cells is crucial for the forming of lymph nodes. The realization that immune cell interaction with stromal cells is central to lymph node formation started another new research field in which the importance of stromal cells for the immune system is studied. Now the different roles that various stromal cell subsets have on the immune response during homeostasis and disease is further dissected using advanced techniques such as single cell RNA sequencing and fluorescence is situ hybridization, in order to discover novel points of interference for targeted therapy in autoimmunity and cancer.
Research Line
Functional development of the immune system
Lymphoid organs are built by a network of stromal cells, which are instrumental for formation of these organs in early ontogeny. Secondary lymphoid organs and their compartments greatly enhance the odds that rare antigen-specific T cells encounter dendritic cells that present the antigen and are thus crucial for effective immune responses. We study this process of lymph node formation and the cellular and molecular mechanisms that are required to form these highly organized lymphoid organs. Special attention is paid to the differentiation requirements of stromal precursor cells to the different stromal subsets that are present in adult lymphoid organs.
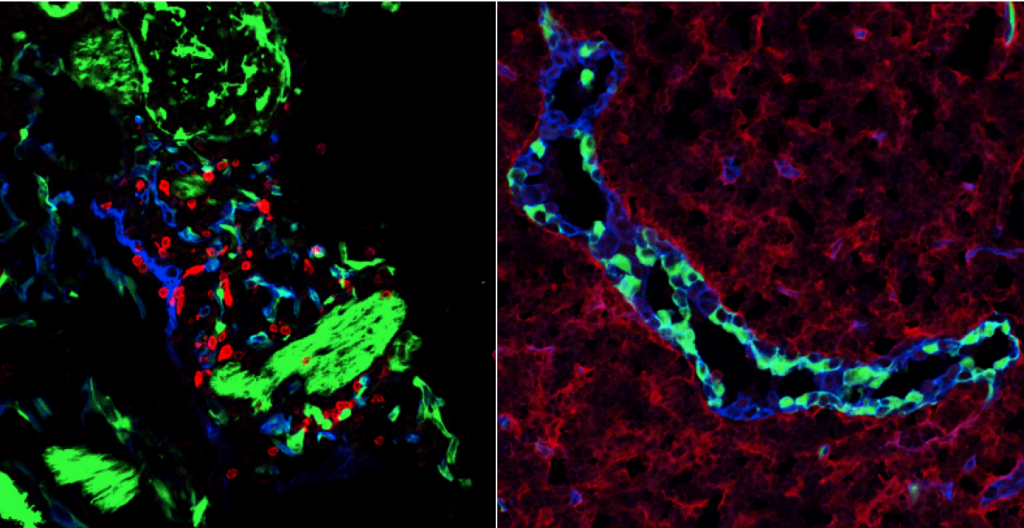
Environmental control of the immune response
Stromal cells within the lymph nodes are able to convey location specific as well as survival signals to immune cells. The formation of lymphoid compartments is not restricted to ontogeny, since ectopic lymphoid structures are formed during chronic inflammatory diseases as well in solid tumours, where they can either contribute to the inflammatory response or an anti-tumour response, respectively. Within the organized structures various different stromal subsets are present, each with their own function. Further characterization of these functions reveal more specifics by which the immune system is controlled by its environment.
In addition, the micro environmental control outside of organized lymphoid organs are studied in inflammatory diseases as well as in cancer in order to better understand how the micro-environment contributes to disease.
Recent key publications
1. Gago da Graça C, van Baarsen LGM, Mebius RE. Tertiary Lymphoid Structures: Diversity in Their Development, Composition, and Role. J Immunol. 2021 Jan 15;206(2):273-281.
2. Nadafi R, Gago de Graça C, Keuning ED, Koning JJ, de Kivit S, Konijn T, Henri S, Borst J, Reijmers RM, van Baarsen LGM, Mebius RE. Lymph Node Stromal Cells Generate Antigen-Specific Regulatory T Cells and Control Autoreactive T and B Cell Responses. Cell Rep. 2020 Mar 24;30(12):4110-4123.
3. Bar-Ephraim YE, Koning JJ, Burniol Ruiz E, Konijn T, Mourits VP, Lakeman KA, Boon L, Bögels M, van Maanen JP, Den Haan JMM, van Egmond M, Bouma G, Reijmers RM, Mebius RE. CD62L Is a Functional and Phenotypic Marker for Circulating Innate Lymphoid Cell Precursors. J Immunol. 2019 Jan 1;202(1):171-182.
4. Bar-Ephraim YE*, Cornelissen F*, Papazian N, Konijn T, Hoogenboezem RM, Sanders MA, Westerman BA, Gönültas M, Kwekkeboom J, Den Haan JMM, Reijmers RM, Mebius RE*, Cupedo T*. 2017. Cross-Tissue Transcriptomic Analysis of Human Secondary Lymphoid Organ-Residing ILC3s Reveals a Quiescent State in the Absence of Inflammation. Cell Rep. 21(3):823-833. * Equal contribution.
5. Koning JJ, Konijn T, Lakeman KA, O’Toole T, Kenswil KJ, Raaijmakers MH, Michurina TV, Enikolopov G, Mebius RE. 2016. Nestin-Expressing Precursors Give Rise to Both Endothelial as well as Nonendothelial Lymph Node Stromal Cells. J Immunol. 197:2686-94.
Group members

Andrew Morrison, MSc
PhD student
My project aims to develop a functional 3D organotypic lymph node with integrated lymphatics that mimic adaptive immune responses. Combining research on lymph node stromal cells and current in-house expertise of tissue engineering & microfluidic devices, the overall objective of the project is to design an organ-on-chip model that features lymphatic drainage of an immune competent gut into a lymph node. The immunosurveillance capability of the model will then be assessed by inducing an inflammatory response, leading to its availability as a future platform for rheumatoid arthritis drug testing.

Daphne Panocha, MSc
PhD student
My work focuses on the development of a human immunocompetent 3D lymph node-on-chip model with integrated lymphatics that mimics adaptive immune responses. We aim to incorporate this 3D lymph node in an organ-on-chip platform by combining it with an immunocompetent skin-on-chip model in a lymph vascularized chip. Functionality of this skin-and lymph node-on-chip model will be assessed by the topical application of contact sensitizers. Important aspects of my project include lymph node stromal cell culture and tissue engineering.

Eelco Keuning, MSc
Lead technician
As lead technician my task is to organize and manage the laboratories at our department so everything runs smoothly. I do this together with our lab manager Ed Döpp. In this management position my role is to focus on quality and safety. In addition I’m the experimental animal research coordinator of the department, for which I function as central contact to all involved parties and can support in many different in vivo models, biotechnical techniques as well as several operations.

Estefany Burniol Ruiz, MD
PhD student
In my research I focus on ILCs in IBD and the changing of homing markers. In 2019 my project became a part of the TIMID consortium. With TIMID we will be able to look at immune-cell alterations in PBMC’s in various immune-mediated diseases. Within the consortium we will be able to combine this with the data from all the other specialists in order to get better insights in these diseases.

Janna Roet, MSc
PhD student
The focus of my project is to unravel the mechanisms of self-antigen expression in lymph node stromal cells, which is needed for the induction of peripheral tolerance. By using a whole-genome CRISPR activation screen we aim to identify the regulatory factors of self-antigen expression. Discovering these regulators will give more insights in fundamental mechanisms of autoimmunity and might be used to design new therapeutic strategies. For this project I make use of flow cytometry, multiplex microscopy, vector cloning and transfections, transductions and RNA sequencing data.

Lotte de Winde, PhD
Research associate
I study the tumor-promoting role of lymph node stromal cells in B-cell lymphoma, and in sentinel lymph nodes draining from solid tumors (melanoma, breast cancer). More specifically, I investigate how interactions between lymph node stromal cells and tumor cells contribute to tumor progression by driving tumor cell dissemination and/or escaping anti-tumor immunity. In my projects, I use primary human lymph node and tumor material as well as cell lines in 2D and 3D co-culture assays, multicolor (spectral) flow cytometry, (advanced) fluorescence microscopy, and molecular and cell biology assays

Mariateresa Coppola, MD, PhD
Postdoctoral researcher
Unrevealing new facets of the human immune responses fascinates me enormously, especially since I believe this knowledge can be translated into a better prevention and treatment of disabling immune-mediated diseases.
Within the TargetToB consortium (Target to B – Target to B (target-to-b.nl)), I want to deep-phenotype B and follicular T-helper cells by mass-cytometry and further characterize newly-discovered cell populations with functional assays. This approach will increase our comprehension of the heterogeneous cell subsets involved in the pathogenesis of auto-immune diseases and cancer which is indispensable to identify novel biomarkers predicting therapy-outcomes and disease-prognosis.

Michael de Kok, MSc
Embedded / Supporting Bioinformatician
My function is to support the entire department with all bioinformatics needs. With so many different projects and data acquisition methods within the department, there is a need for a dedicated person to develop, maintain and teach different scripts and pipelines for all data analyses. This can be transcriptomics, proteomics, imaging or any other conceivable pipeline. People can approach me for assistance with their analyses and quickly get their current problems solved. In addition I manage data, e-lab journals and co-host multiple meetings and courses concerning bioinformatics as well as teaching subjects such as programming and good data practices.

Tanja Konijn, Ing
Research technician
I work together with Postdocs and AIO’s on several projects concerning lymph development and mucosal immunology. Since 2014 I became, in combination with my function as technician, an operator at the Microscopy & Cytometry Core Facility (MCCF).










