Research Themes
The research performed in the department of Molecular Cell Biology and Immunology covers a wide variety of topics in the field of cell biology and immunology in health and disease.
Our research approaches vary from sophisticated molecular, biochemical, and cellular methods to high dimensional cytometry, microscopy, using engineered organotypic models, animal models, to apply knowledge for clinical translation (diagnostic or therapeutic). In particular, basic questions relevant for cancer, immunodeficiencies, inflammatory bowel diseases, rheumatoid arthritis, allergy, skin diseases, systemic lupus erythematosus, multiple sclerosis, Alzheimer disease and vascular diseases are being addressed.
Cancer Immunology
Aim
The overall goal of our translational research in the field of Cancer Immunology is to investigate new immunotherapy’s by vaccination and antibody therapy. We focus on both the tumor specific innate and the adaptive immunity overcoming the tumor suppressive tumor micromilieu. We develop new multiparameter immune monitoring panels and analyse novel immune suppressive molecular players within the tumor microenvironment to interfere with to further improve immune diagnostics and the design of novel immunotherapy’s. Our focus is on melanoma, colonrectal and pancreatic cancer and glioblastoma.
Approach
We use in vitro and in vivo models for melanoma, colon, pancreatic cancer and glioblastoma. We design and synthesize DC targeting nanovaccines modified peptidenetworks, and have multiparameter parameter CYTOF analysis and microscopy analysis and firefly imaging systems to analyse immunological responses. We use antibody treatments in the metastatic formation of tumors. Moreover using CRISPR-Cas we modify regulatory glycosylation mechanisms in tumor to study their effects on tumorimmunity. One of our major strengths is that we have access to well-characterized patients samples that allowing us to translate our fundmental findings to clinical applications.
Principal Investigators
Prof. dr. Yvette van Kooyk studies DC targeted cancer vaccines and combination therapies aimed to redirect immune regulatory programs in the tumor microenvironment that are caused by the glyco-code.
Prof. dr. Reina Mebius focusses on the role of stromal cells in the survival and maintenance of tumor cells, as well as their effect on lymphocytes in relation to anti-tumor immunity.
Prof. dr. Marjolein van Egmond investigates novel antibody formats to engage immune effector cells in tumor cell elimination and to initiate anti-tumor immune responses.
Dr. Joke den Haan investigates the capacity of different types of myeloid antigen presenting cells (APCs) to activate anti-cancer immune responses and develops cancer nanovaccines that are delivered to specific APC cell types.
Dr. Sandra J. van Vliet dissects how aberrant glycan signatures in the tumor microenvironment signal through glycan-binding lectin receptors to alter anti-tumor immunity.
Dr. ir. Jan Van den Bossche investigates macrophage immunometabolism and aim to understand the mechanisms by which metabolic crosstalk in the tumor microenvironment affects macrophage function, thereby uncovering new strategies to manage disease outcome.
Dr. Juan J. Garcia Vallejo uses high dimensional cytometry methods to characterize the immune composition both in tumors and blood in several cancer models with the aim of identifying common mechanisms of disease and cellular biomarkers.
Dr. Febe van Maldegem investigates how the different cells in the tumour microenvironment form complex cellular networks in space and time, and how we can use that to strengthen immune responses in therapy.
Mucosal Immunology
Aim & Approach
Mucosal surfaces (i.e. of the intestines, the lungs and urogenital tract) represent a vast interface that shields the interior of the body from external influences. In healthy circumstances, the mucosal immune system protects against pathogens, while at the same time it must maintain tolerance towards innocuous inhaled or ingested antigens and commensal micro-organisms. In chronic inflammatory bowel disease, the immune system is overly active. In allergies, first exposure to an allergen via the oral mucosa can lead to tolerance rather than delayed type IV hypersensitivity. In the theme ‘Mucosal Immunology’, we investigate normal and abnormal development of mucosal immune responses.
Principal Investigators
Prof.dr. Reina Mebius focusses on the role of stromal cells in lymphoid organ development and the mucosal immune system, as well as on the vitamin A metabolite retinoic acid (RA), which is instrumental in regulating gut associated immune responses.
Prof.dr. Marjolein van Egmond addresses the function of immunoglobulin A (IgA), which is the prevalent antibody present in mucosal areas, and the effector mechanisms that are triggered via the IgA Fc receptor FcαRI.
Prof.dr. Sue Gibbs uses state of the art tissue engineered oral mucosa, gut, lymph node organotypic models to investigate tolerance and innate immune mechanisms.

Neuroimmunology
Aim & Approach
The majority of neurological disorders are either caused or frequently accompanied by alterations in the immune system, resulting in chronic neuroinflammation. Research in the neuro-immunology group focuses on the understanding of how altered immunological and inflammatory effects contribute to the progression of neurological diseases. Specifically, we study how cellular alterations in the immune system, at the brain barriers and in the glial compartments underlie neuroinflammatory and neurodegenerative conditions. Together with our (pre)clinical partners of the MS center Amsterdam, Alzheimer Center Amsterdam and stroke unit within Amsterdam UMC, we apply a translational approach to investigate these pathways in disorders, such as multiple sclerosis (MS), stroke and Alzheimer disease (AD). Our ultimate goal is to define novel therapeutic ways to resolve the detrimental effects of chronic neuroinflammation.
Principal Investigators
Prof. dr. Elga de Vries focuses on the role of understanding of the cross talk of the immune system and brain to fight neurological diseases with specific attention for the role of the brain barriers therein.
Dr. Gijs Kooij’s major aim is to understand the natural process to resolve neuro-inflammation in order to provide new perspectives on disease pathogenesis and to reveal new diagnostic & treatment opportunities.
Prof. dr. Jack van Horssen has expertise in histopathological analysis of brain specimens.
Skin Immunology
Aim & Approach
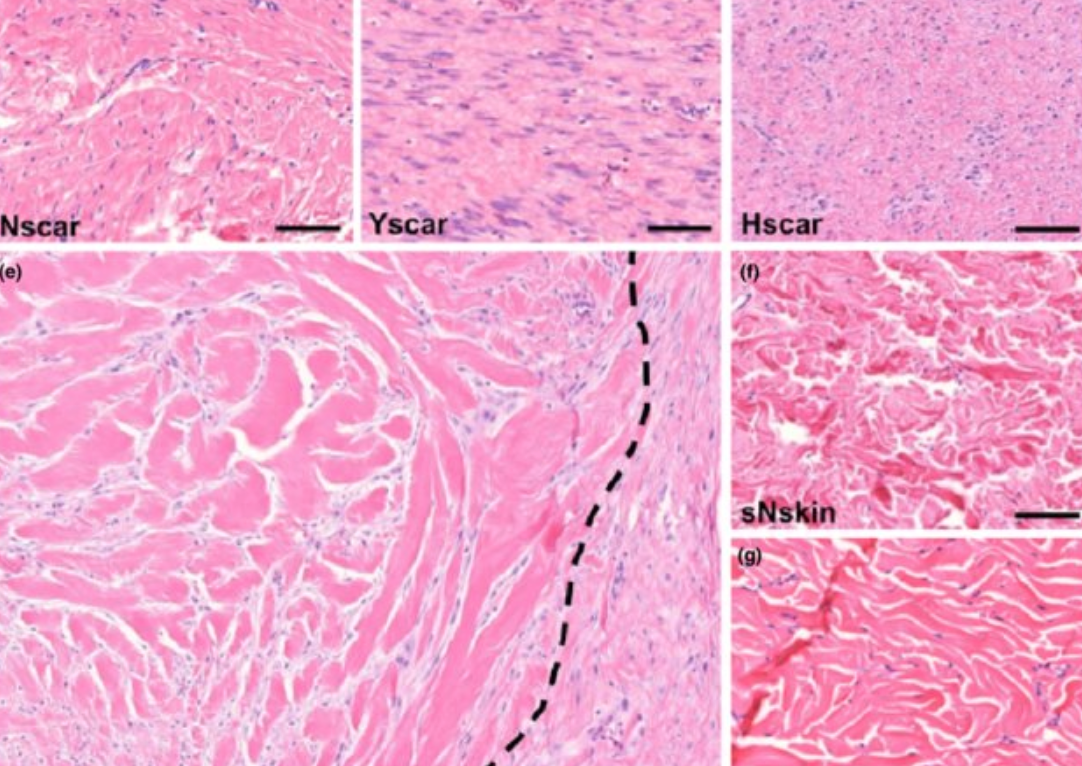
Our skin functions as a complex physical barrier between the environment and our internal organs. It is our 1st line of defense for pathogens and substances coming into contact with the skin. In the healthy situation, various innate and adaptive immune mechanisms are activated to prevent pathogens and toxic substances entering the systemic circulation where they can adversely influence other organs. When this skin immunity malfunctions, skin disease can occur. In the theme ‘Skin Immunology’, we investigate normal and abnormal development of skin immune responses. Moreover, we study how dendritic cell targeting vaccines, modulate skin resident dendritic cells to alter T cell responses.
Principal Investigators
Prof.dr. Sue Gibbs focuses on inflammatory skin disease and wound healing. She develops state of the art tissue engineered organotypic models to address research questions including immune competent skin-, gingiva-, gut,- lymph node- and melanoma-models and in particular ‘’organ-on-a-chip’’.
Prof.dr. Yvette van Kooyk focusses on the dendritic cell targeting nano-vaccines that enhance the stimulation of T cell responses in cancer or induce regulator T cells to inhibit inflammatory response in allergy.
Immunodeficiencies and disorders of immune dysregulation
Aim & Approach
We study diseases caused by dysregulation of the immune system, including immune deficiencies, immune-mediated disorders such as systemic lupus erythematosus (SLE), rheumatoid arthritis, cardiovascular disease (CVD), as well as allergies and asthma. Our aim is to improve understanding of molecular and immunological mechanisms leading to these diseases and to develop novel diagnostic and therapeutic approaches.We use extensive immune cell phenotyping, genomic, transcriptomic, metabolomic and glycomic approaches to analyse various disorders of immune dysregulation, discover novel immunological biomarkers and develop new personalised therapies.
Principal Investigators
Prof. dr. Sergey Nejentsev focuses on primary immunodeficiencies and mechanisms of susceptibility to infection and characterises novel biomarkers in SLE patients.
Prof. dr. Reina Mebius focusses on the role of lymph node stromal cells in controlling adaptive immune responses during homeostasis and chronic inflammatory responses such as rheumatoid arthritis.
Dr. Jan Van den Bossche investigates how dysregulated metabolism drives aberrant macrophage activation and inflammation, with the goal to identify new treatment options for chronic inflammatory diseases such as atherosclerosis.
Prof. dr. Yvette van Kooyk develops novel therapeutic approaches aiming to induce tolerance in patients with allergy and asthma
Dr. Juan J. Garcia Vallejo is interested in how the immune system is altered in situations so different as exposure to microplastics or infection by the SARS-CoV-2 virus.
Dr. Sandra J. van Vliet aims to develop small molecules targeting C-type lectins to manipulate and employ their signaling and immunomodulatory capacities to correct immune disorders.